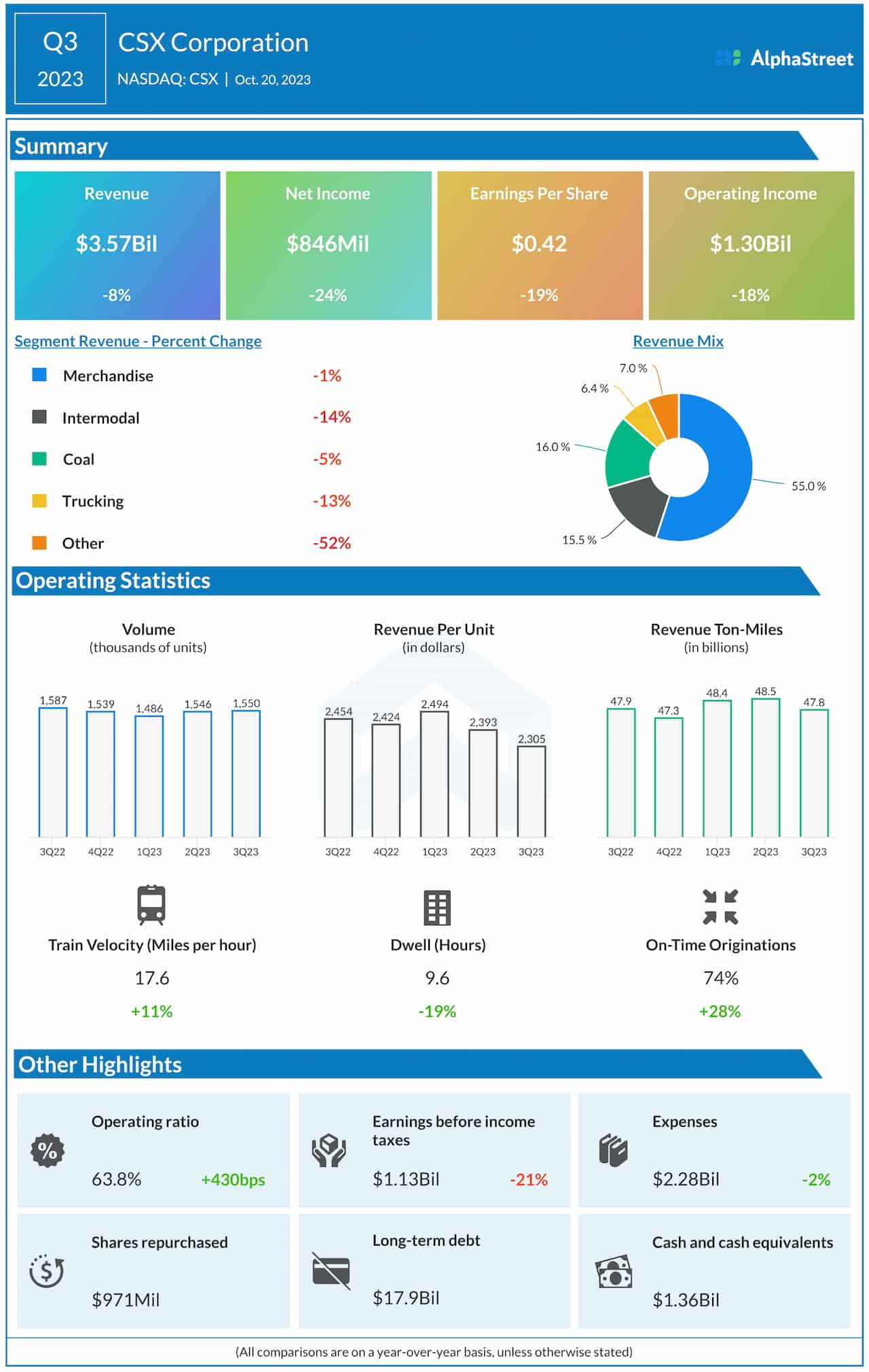
Amgen (NASDAQ:AMGN) announced Friday that its Phase 2 DeLLphi-301 study for bispecific T-cell engager tarlatamab showed an objective response rate of 40% in a Phase 2 trial for patients with small cell lung cancer.
The potentially registrational DeLLphi-301 was designed to evaluate tarlatamab in SCLC patients who had failed two or more prior lines of treatment.
The ORR, the trial’s primary endpoint, was based on 100 patients who received tarlatamab at the selected 10 mg dose with a median follow-up of 10.6 months.
Regarding key secondary goals, median progression-free survival and median overall survival reached 4.9 months and 14.3 months, respectively, while median response duration was not reached, Amgen (AMGN) said.
As for safety, 4% of patients discontinued the trial due to treatment-related adverse events (TRAE), while cytokine release syndrome (CRS) and pyrexia were among the most frequent treatment-emergent adverse events in the tarlatamab 10 mg group.
The company is scheduled to present the results at a medical event in Madrid, Spain, later today.
A Phase 3 trial called DeLLphi-304, designed to test the candidate as a second-line option for SCLC against standard-of-care chemotherapy, is currently enrolling patients. Plans to conduct two additional late-stage trials for tarlatamab in earlier lines of settings for SCLC are also underway.
A previous readout indicated a 23% ORR for tarlatamab in a Phase 1 dose exploration and expansion study involving SCLC patients.